Major Research Areas
Overview
Our research program, focused on studying nucleic acids–ligand interactions, developing new emissive nucleosides and oligonucleotides, and advancing novel cellular delivery agents, brings together synthetic chemistry, biophysics, molecular biology and cell biology.
It provides a multidisciplinary education and training ground for students, with emphasis on biophysical techniques that illuminate the stability, folding, and recognition features of biopolymers.
One of the main goals of our early efforts was to uncover the fundamental recognition elements that govern RNA–small molecule interactions and to utilize them for the development of new RNA targeting ligand as cellular effectors as well as antibacterial and antiviral agents.
Relevant references:
RNA and the Small Molecule World. Y. Tor, Angew. Chem. Int. Ed. 1999, 38, 1579–1582.
A Novel Solid-Phase Assembly for Identifying Potent and Selective RNA Ligands. N. W. Luedtke and Y. Tor, Angew. Chem. Int. Ed. 2000, 39, 1788–1790.
Targeting RNA with Small Molecules Y. Tor, ChemBioChem. 2003, 4, 998-1007.
Fluorescent Nucleosides

We focus on the design, synthesis and photophysical evaluation of new isomorphic fluorescent nucleoside analogs, as well as on their utilization for the fabrication of “real-time” fluorescence-based discovery and biophysical assays.
Relevant references:
Simple Fluorescent Pyrimidine Analogs Detect the Presence of DNA Abasic Sites N. J. Greco and Y. Tor, J. Am. Chem. Soc. 2005, 127, 10784-10785.
Emissive RNA Alphabet D. Shin, R. W. Sinkeldam and Y. Tor, J. Am. Chem. Soc. 2011, 133, 14912–14915.
Enzymatic Interconversion of Isomorphic Fluorescent Nucleosides: Adenosine Deaminase Transforms an Adenosine Analogue into an Inosine Analogue R. W. Sinkeldam, L. S. McCoy, D. Shin, Y. Tor. Angew. Chem. Int. Ed. 2013, 52, 14026 –14030.
Cellular Delivery

We focus on the development of guanidinium-rich cellular delivery agents and investigate the mechanistic aspects of their cellular uptake and their potential therapeutic applications.
Relevant references:
Guanidinylated Neomycin Delivers Large, Bioactive Cargo into Cells through a Heparan Sulfate-dependent Pathway L. Elson-Schwab, O. B. Garner, M. Schuksz, B. E. Crawford, J. D. Esko, and Y. Tor,Biol. Chem. 2007, 282, 13585–13591.
On Guanidinium and Cellular Uptake E. Wexselblatt, J. D. Esko, and Y. Tor, J. Org. Chem., 2014, 79, 6766–6774.
GNeosomes: Highly Lysosomotropic Nanoassemblies for Lysosomal Delivery E. Wexselblatt, J. D. Esko, and Y. Tor, ACS Nano, 2015, 9, 3961–3968.
Prebiotic Chemistry
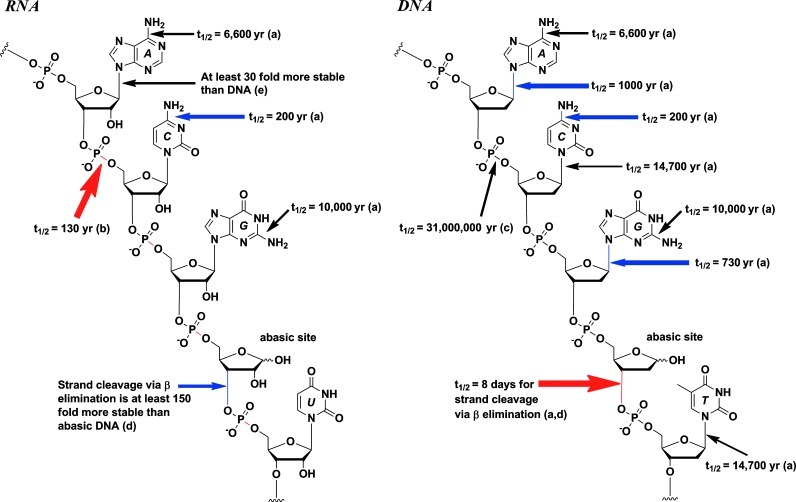
We are interested in the plausible selection pressures and processes that have contributed to the evolutionary fitness of the contemporary genetic alphabet.
Relevant references:
Refining the genetic alphabet: A late period selection pressure? A. C. Rios and Y. Tor, Astrobiology, 2012, 12, 884–891.
On the origin of the canonical nucleobases: an assessment of selection pressures across chemical and early biological evolution A. C. Rios and Y. Tor, Israel J Chem 2013, 53, 469–483.
Hydrolytic stability of N-glycosyl bonds in modified, alternative and damaged nucleosides: Selection pressure for the genetic alphabet A. C. Rios, H. T. Yu, Y. Tor, J. Phys. Org. Chem. 2015, 28, 173-180.
